 Several recent studies have incorporated Bayesian parameter estimation and uncertainty quantification (UQ), however, most of these studies ignore the model mismatch. In addition, we estimate micro-circulation parameters by introducing global scaling factors for the boundary condition parameters at the terminal arteries. We investigate if the vessel stiffness is (a) constant over the entire network (as suggested in ), (b) increases with decreasing radius (as suggested in ), or (c) should be estimated for each vessel separately. We compare two constitutive equations: a linear and a nonlinear wall model relating vessel pressure and area. We examine several alternative models to select the model that best predicts the data, which is performed using statistical model selection criteria based on the Watanabe Akaike information criterion (WAIC). The current study focuses on inferring and analysing parameters intrinsic to the haemodynamic model, including the vessel stiffness and parameters specifying the micro-circulation (boundary conditions via three-element Windkessel models attached at the terminal vessels), in a fixed network. Similar to previous studies, the 1D models analysed here have two types of parameters: specifying the vessel network (radius, length and the connectivity of arteries) and the haemodynamics (pressure and flow). Our work shows that the consequence of ignoring model discrepancy is biased haemodynamic predictions and parameters, and thus inability of reliably using these models in the clinic. While these investigations provided valuable insight into the physiology, they ignored an essential source of uncertainty resulting from the inadequacy of the model form. These studies minimize the least-squares error between the model output and the measurements. However, only a few have aimed at devising subject-specific predictions by estimating model parameters. Several previous studies have developed 1D fluid-dynamics models predicting pulmonary blood flow and pressure.  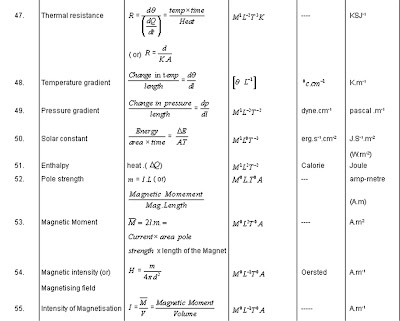
the errors from numerically integrating the model equations). Our analysis includes the uncertainty in the model parameters (which are naturally variable), in the model form/structure (the discrepancy between the model and the reality), in the measurements (the noise model), and in the simulator output (e.g. We highlight the importance of determining the uncertainty when calibrating the model to the data. Predictions of blood pressure, blood flow and vessel area are computed in an arterial network model constructed from micro-CT images from a control mouse, and the pressure predictions are compared to dynamic data in the main pulmonary artery (MPA). The current study assesses the health of the pulmonary system by integrating imaging data (obtained with micro-computed tomography (CT)), blood pressure data (measured invasively via catheterization) and blood flow data (measured with ultrasound), using a one-dimensional (1D) fluid-dynamics model combined with statistical inference. Before using the models for decision-making in the clinic, they must be calibrated and fitted to data, and their credibility rigorously tested by modelling all sources of uncertainty using statistical analysis. The ultimate goal is achieving personalized medicine, to allow patient-specific care and treatment. Results show that the nonlinear pressure–area relationship with stiffness dependent on the unstressed radius predicts best the data measured in a control mouse.Ĭomputational haemodynamics models are emerging as powerful tools for analysing cardiovascular disease progression and the effects of treatments by providing essential haemodynamic metrics which could not be obtained from in vivo experiments. We use formal model selection analysis based on the Watanabe Akaike information criterion to select the model that best predicts the pulmonary haemodynamics. Additionally, we compare a linear and a nonlinear wall model, as well as models with different vessel stiffness relations. We show that our proposed method allowing for model mismatch, which we represent with Gaussian processes, corrects the bias. We demonstrate that minimizing the mean squared error between the measured and the predicted data (the conventional method) in the presence of model mismatch leads to biased and overly confident parameter estimates and haemodynamic predictions. We emphasize an often neglected, though important source of uncertainty: in the mathematical model form due to the discrepancy between the model and the reality, and in the measurements due to the wrong noise model (jointly called ‘model mismatch’).  
This study uses Bayesian inference to quantify the uncertainty of model parameters and haemodynamic predictions in a one-dimensional pulmonary circulation model based on an integration of mouse haemodynamic and micro-computed tomography imaging data. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
